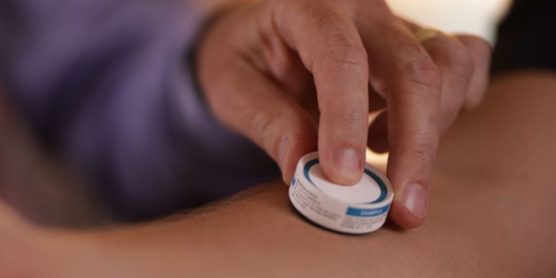
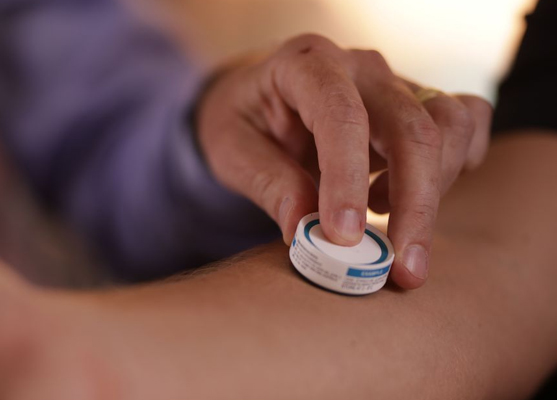
Clinical trial paves way for “micro patch” vaccine delivery
Vaxxas is pioneering a next-generation vaccine delivery platform that targets immunological cells below the surface of the skin.
Today, the company published the results of its Phase I clinical trial in the PLoS Medicine journal revealing that its high-density microarray patch (HD-MAP) has shown great potential to deliver an enhanced immune response, quicker and at a lower dose than conventional methods.
During the clinical trial involving 210 people who were administered the flu vaccine, researchers found that vaccine delivered through the patch, which has a surface with thousands of “micro-projections” just a quarter of a millimetre long, could produce six times the immune response as the same amount of vaccine delivered into the bloodstream via needle and syringe, and more quickly. Thus, by enabling faster and more effective immune response, the patch could establish protection from infection in days instead of weeks, which is a significant public health benefit and extremely important for both seasonal and pandemic influenzas.
The publication also highlighted that vaccine patch was stable when stored at 40°C (104°F) for at least 12 months, providing the potential for easy distribution without the cost and complexity of continuous refrigeration.
Vaxxas also announced that it has received US$ 5 million (AU$ 7.5 million) in funding from the Bill & Melinda Gates Foundation to conduct initial human clinical studies using its “micro patch” for measles and rubella vaccination.